Beskrivning
# Is Red Light Therapy Safe For Breast Cancer Patients In Switzerland
## Introduction
Red light therapy (RLT), also called photobiomodulation (PBM) or low-level laser therapy (LLLT), uses low-intensity red or near-infrared light to stimulate cellular repair and reduce inflammation. For breast cancer patients in Switzerland, curiosity about RLT often ties to managing treatment side effects—like radiation-induced skin damage, lymphedema, or fatigue. However, the safety of RLT for this population remains a critical question, as concerns about tumor growth or interference with standard care persist. This article explores current research, Swiss medical guidelines, and key considerations for patients evaluating RLT.
## Current Research on RLT and Breast Cancer
### Global and Swiss Study Findings
Most RLT research on breast cancer focuses on side effect management in survivors, not active treatment. A 2022 systematic review in the *Journal of Clinical Oncology* noted small studies found RLT may reduce lymphedema severity in survivors, but larger randomized controlled trials (RCTs) are needed to confirm results.
In Switzerland, a 2023 University of Zurich oncology study found RLT alongside standard care reduced radiation-induced skin toxicity in 30 breast cancer patients—all of whom had completed active chemotherapy and were in survivorship. No Swiss studies have evaluated RLT safety in patients with active breast cancer (e.g., those undergoing chemotherapy or radiation).
A key gap is data on RLT’s effect on tumor biology. Lab studies suggest red light may stimulate angiogenesis (new blood vessel growth), which could theoretically support tumor growth—but these findings have not been replicated in human trials.
## Swiss Medical Guidelines & Expert Perspectives
### Complementary Therapy Guidance
The Swiss Cancer League (Ligue Suisse contre le Cancer) advises all complementary therapies (including RLT) be discussed with an oncologist before use. This ensures no interactions with standard treatments or risks to active disease.
The Swiss Society of Oncology (SSO) echoes this: while complementary therapies can support well-being, they never replace evidence-based care. For RLT, the SSO states insufficient evidence exists to recommend it during active breast cancer treatment, as potential tumor stimulation risks are unruled out.
The Federal Office of Public Health (FOPH) regulates medical devices via Swissmedic. Only Swissmedic-approved RLT devices are safe for clinical use—over-the-counter devices without certification are not recommended for breast cancer patients.
## Potential Risks for Breast Cancer Patients
### Unique Safety Concerns
RLT is low-risk for healthy individuals, but breast cancer patients face distinct considerations:
1. **Tumor Progression**: Theoretical angiogenesis risks (from lab studies) mean active cancer patients should avoid RLT until more data exists.
2. **Hormone Receptor Effects**: 70% of breast cancers are estrogen receptor (ER)-positive. Preliminary lab studies suggest RLT may affect ER activity, but clinical data is inconsistent.
3. **Treatment Interference**: RLT stimulates cellular repair, which could counteract chemotherapy/radiation (designed to kill rapidly dividing cells). This is theoretical but a key concern.
4. **Unregulated Devices**: Non-Swissmedic-approved devices may emit incorrect wavelengths, causing skin burns or eye damage—especially risky for patients with radiation-compromised skin.
## Safe Use Considerations for Survivors
### Conditions for RLT in Survivorship
For survivors (no active disease for ≥1 year), RLT may be considered only if:
1. **Medical Clearance**: Written approval from the oncologist or multidisciplinary team (to avoid interference with surveillance/hormone therapy).
2. **Swissmedic-Approved Devices**: Only devices tested for intended use (e.g., lymphedema management) are allowed.
3. **Targeted Application**: Applied to specific areas (e.g., lymphedema-affected arm) — never over the original tumor site or suspected recurrence.
4. **Supervised Use**: Initial sessions by trained professionals (physical therapists/oncology nurses) to ensure correct dosage.
5. **Monitoring**: Immediate reporting of changes (pain, swelling, new lumps) to the care team.
## Personalized Care in Switzerland
### Individualized Assessment
Switzerland’s healthcare system emphasizes personalized cancer care. Each patient’s case (tumor type, stage, treatment history) dictates RLT safety:
– A survivor with early-stage ER-positive cancer (no active disease) may qualify for RLT for lymphedema, after thorough review.
– A metastatic patient on active chemotherapy would likely be advised against RLT due to theoretical risks.
Many Swiss oncology centers offer integrative medicine services, where patients discuss complementary therapies with specialists aligned with their care team.
## Conclusion
Red light therapy is not recommended for breast cancer patients during active treatment in Switzerland due to insufficient safety data. For survivors, RLT may be considered for specific side effects only under strict conditions: medical clearance, Swissmedic-approved devices, supervised use, and monitoring.
Complementary therapies never replace standard care. Any RLT decision must involve the multidisciplinary team to protect patient health. As research progresses, more data may clarify RLT’s role—but for now, Swiss guidelines prioritize caution and personalized assessment.
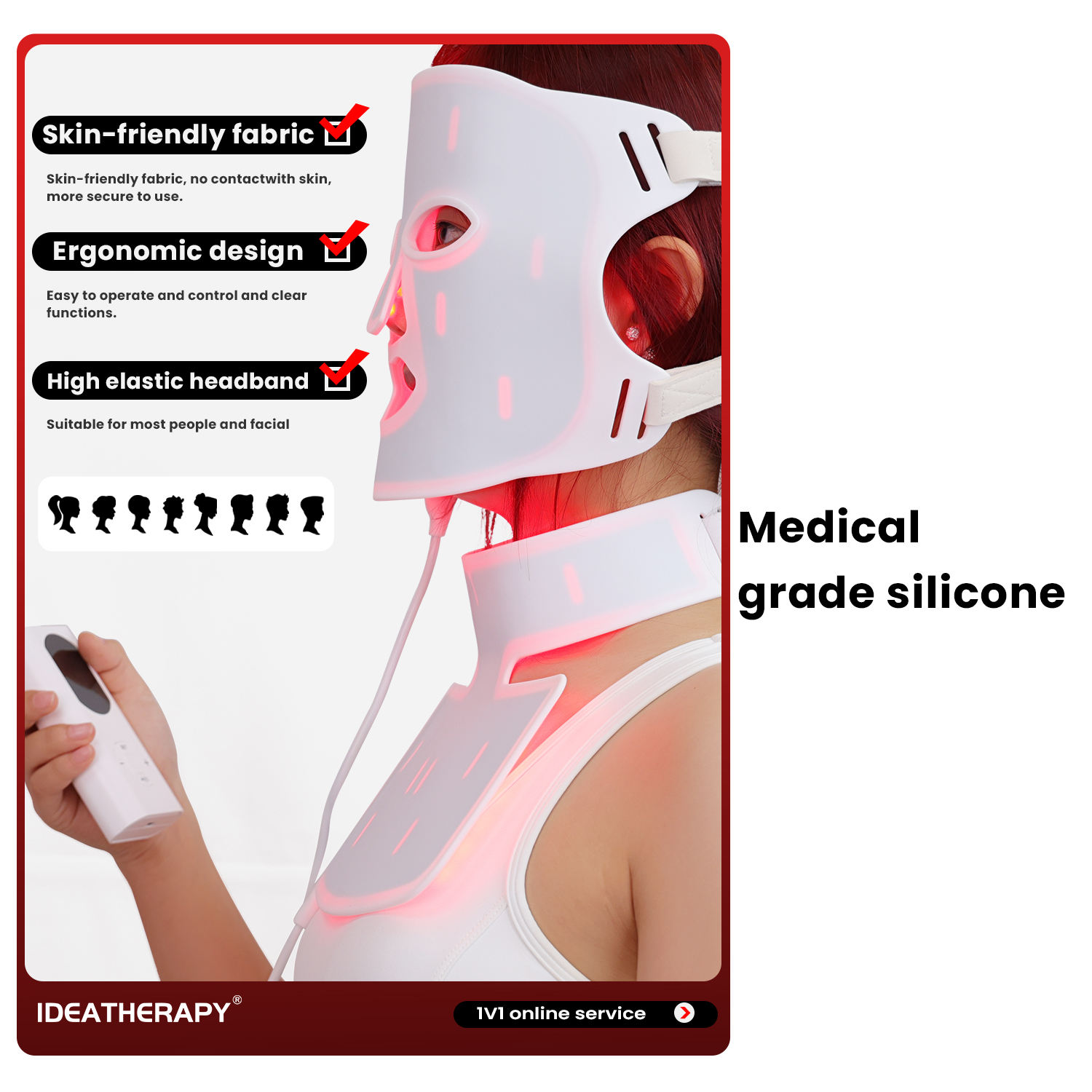


Recensioner
Det finns inga recensioner än.